Pakistan, India post uptick in coronavirus cases
NIH data shows daily Covid tally rose by 25 cases, in India by 2,000 infections: Morocco refuses entry to passengers on Covid-hit liner: Moderna announces positive results for Omicron vaccine
June 9, 2022 10:39 AM

There was an uptick in everyday coronavirus infections in Pakistan and India; however there was no fatality reported in Pakistan while the Indian authorities registered another eight deaths during the last 24 hours (Wednesday).
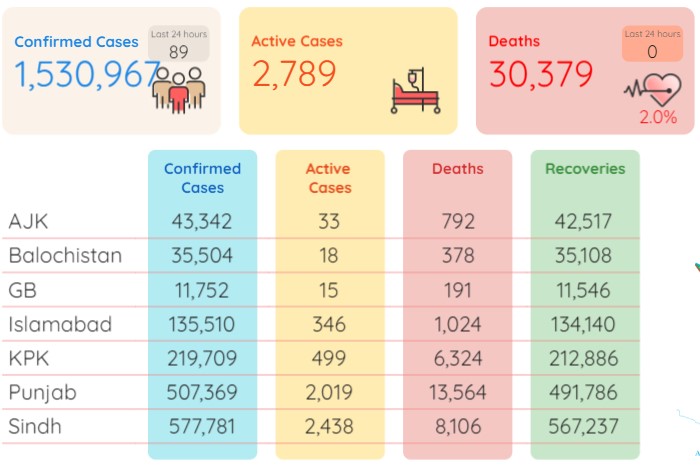
Pakistan recorded 89 coronavirus cases 25 more than posted a day earlier while in neighbouring India the situation is becoming serious as it registered over 7,000 cases which were about 2,000 more as compared to the previous single-day tally.
According to the latest figures released by the National Institute of Health Pakistan on Thursday morning, the death toll remained the same at 30,379, whereas the number of total infections now stood at 1,530,967 after adding the fresh 89 cases.
During the last 24 hours (Wednesday), 15,178 tests were conducted throughout Pakistan whereas the positivity ratio stood at 0.59 percent. The number of patients in critical care was recorded at 44.
https://twitter.com/NIH_Pakistan/status/1534716290241282049
During the last 24 hours (Wednesday), another 55 people recovered from the Covid-19 and the number of total recoveries now stood at 1,497,799. As of Thursday, the total count of active cases in the country was recorded at 2,789.
As many as 577,781 coronavirus cases have so far been confirmed in Sindh, 507,369 in Punjab, 219,709 in Khyber Pakhtunkhwa, 135,510 in Islamabad, 35,504 in Balochistan, 43,342 in Azad Kashmir and 11,752 in Gilgit-Baltistan.
As many as 13,564 individuals have lost their lives to the pandemic in Punjab so far, 8,106 in Sindh, 6,324 in KP, 1,024 in Islamabad, 792 in Azad Kashmir, 378 in Balochistan and 191 in Gilgit Baltistan.
India reports over 7,000 Covid-19 cases
India on Thursday reported a total of 7,240 new coronavirus infections, taking the overall tally of ovid-19 cases in the country to 4,31,97,522, the Health Ministry said. The daily positivity rate of 1.62 percent was observed in the last 24 hours, while the weekly positivity rate was recorded to be 0.91 percent, the Indian Health Ministry said.
The active Covid-19 cases rose to 32,498 and the number of deaths due to Covid climbed to 5,24,723 with eight deaths in the last 24 hours, according to the Indian government data.
Morocco refuses entry to passengers on Covid-hit liner
Morocco has refused entry to passengers on a German cruise liner after some tested positive for Covid-19, Moroccan media reported Wednesday.
The Mein Schiff Herz, owned by German tourism giant TUI and capable of carrying 1,900 passengers plus crew, had been meant to dock in Tangiers on the Gibraltar Straits on Tuesday morning on its way to Lisbon.
But Moroccan authorities barred passengers from disembarking due to "a report received by the Moroccan health authorities which indicates an increase in cases of Covid-19 infection among passengers," according to news website Le360.
Morocco on Tuesday raised its alert level for Covid-19 from green to orange after recorded cases spiked in several major cities.
Death and hospitalisation figures are relatively low but the rising caseload comes as Morocco's tourism operators -- hit by two years of near-zero visitor numbers -- desperately hope for a recovery.
Last month the kingdom scrapped rules that had obliged incoming travellers to present a negative PCR test.
Moderna announces positive results for Omicron vaccine
US biotech company Moderna on Wednesday announced positive results for a new vaccine that targets both the original Covid strain and Omicron, and sees the shot as its "lead candidate" for a booster this fall.
The so-called "bivalent" vaccine was tested in a trial of 814 adults and shown to produce 1.75 times more Omicron-specific neutralizing antibodies, which have the power to prevent infection, compared to Moderna's original Spikevax vaccine.
All of the participants previously received three doses of Spikevax, and then slightly more than half went on to get a fourth dose of the bivalent shot while the rest got another dose of Spikevax. Antibody levels were tested one month later.
The group that got the new shot also received slightly superior protection to the ancestral strain of Covid compared to Spikevax -- though original Covid has long since disappeared from circulation.
"We are thrilled," said Stephane Bancel, CEO of Moderna in a statement, adding he anticipated this vaccine would be the company's lead candidate for authorization as a booster this fall.
"We want to be as ready as early as August for shipping," he added to investors in a call.
The results were broadly welcomed by experts, who agreed Moderna's vaccine is now the front runner as a booster, though some gave a note of caution.
"We won't know about clinical outcomes until later this year," tweeted Eric Topol, a physician and scientist at Scripps Research Translational Institute. The study concerns only antibody levels, which are thought to serve as a useful proxy for how a vaccine will perform, but can't make precise predictions.
Breakthrough infections have risen since Omicron became dominant in late 2021, with vaccine makers hoping to restore efficacy to previous levels, even as first generation vaccines continue to protect well against severe disease and death.
- Omicron subvariants challenge -
Moderna officials did concede during the investor call that antibody levels would be lower against Omicron's subvariants that are now in circulation, but said it believed it was still a superior booster than repeating Spikevax.
The BA.2.12.12 variant is currently dominant, followed by BA.2, with BA.4 and BA.5 rising, according to Centers for Disease Control and Prevention (CDC) data. Each successive Omicron subvariant appears to have a transmission advantage over those that came before it.
Moderna also doesn't yet have data on durability -- how the new vaccine booster will fare three months and six months out.
The problem of an ever-evolving virus poses a challenge for health authorities. A panel of Food and Drug Administration experts will meet June 28 to discuss considerations and strategies for boosters in fall and winter.
"Society is moving toward a new normal that may well include annual Covid-19 vaccination alongside seasonal influenza vaccination," top FDA officials wrote in a letter to the Journal of the American Medical Association in May.
President Joe Biden's government has said it has enough funds to deliver additional boosters for those at highest risk this fall -- including the elderly and those with immune compromised conditions.
But whether these vaccines will be made widely available to the general public for free could depend on whether Congress authorizes $22.5 billion in funding the White House is seeking.
With inputs from AFP.






 Maryam Nawaz can wear uniform!
Maryam Nawaz can wear uniform! Here is all about Madiha Rizvi’s second husband
Here is all about Madiha Rizvi’s second husband Madiha Rizvi ties the knot again
Madiha Rizvi ties the knot again Zara Noor Abbas inspired by Rani Mukerji
Zara Noor Abbas inspired by Rani Mukerji Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital
Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital Humayun Saeed and Saboor Aly under fire for close interaction in public
Humayun Saeed and Saboor Aly under fire for close interaction in public





