All indicators up as daily coronavirus cases soar past 300 in Pakistan
NIH data shows positivity ratio builds to 2.22 , active cases tally at 3,797: Study says Covid vaccines saved 20 million lives in first year: EU watchdog approves Valneva anti-Covid jab
June 24, 2022 10:37 AM

Almost all the coronavirus indicators have shown upward trajectory in Pakistan as the country registered another surge in its everyday Covid-19 infections.
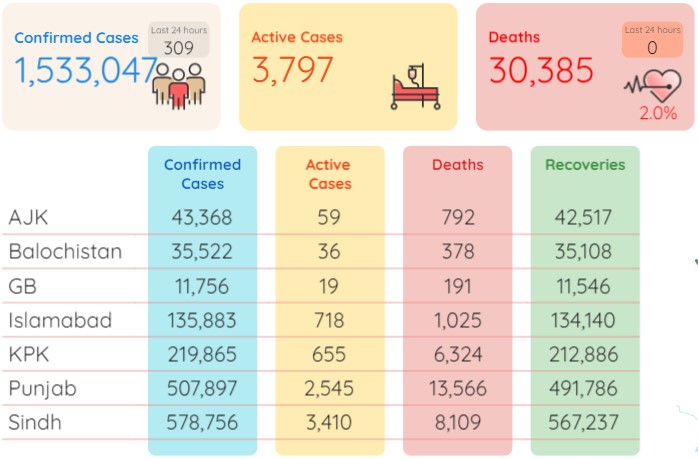
According to the figures released by the National Institute of Health Pakistan on Friday morning, Pakistan has recorded a massive rise in its daily Covid-19 cases as it posted 309 infections with positivity ratio swelling to 2.22 percent during the last 24 hours (Thursday).
As per the NIH data, the death toll in Pakistan remained the same at 30,385 as no fatality was reported from across Pakistan whereas the number of total infections now stood at 1,533,047 after adding the fresh 309 cases.
During the last 24 hours (Thursday), 13,941 tests were conducted throughout Pakistan whereas the positivity ratio also shot up to 2.22 percent. The number of patients in critical care was recorded at 80.
https://twitter.com/NIH_Pakistan/status/1540142785952727043
During the last 24 hours (Thursday), another 167 patients recovered from the Covid-19 in Pakistan and the number of total recoveries now stood at 1,498,865. As of Friday, the total count of active cases in the country was recorded at 3,797.
As many as 578,756 coronavirus cases have so far been confirmed in Sindh, 507,897 in Punjab, 219,865 in Khyber Pakhtunkhwa, 135,883 in Islamabad, 35,522 in Balochistan, 43,368 in Azad Kashmir and 11,756 in Gilgit-Baltistan.
As many as 13,566 individuals have lost their lives to the pandemic in Punjab so far, 8,109 in Sindh, 6,324 in KP, 1,025 in Islamabad, 792 in Azad Kashmir, 378 in Balochistan and 191 in Gilgit Baltistan.
Covid vaccines saved 20 million lives in first year: study
Covid vaccines prevented nearly 20 million deaths in the first year after they were introduced, according to the first large modelling study on the topic released Friday.
The study, published in The Lancet Infectious Diseases, is based on data from 185 countries and territories collected from December 8, 2020 to December 8, 2021.
It is the first attempt to estimate the number of deaths prevented directly and indirectly as a result of Covid-19 vaccinations.
It found that 19.8 million deaths were prevented out of a potential 31.4 million deaths that would have occurred if no vaccines were available.
It was a 63 percent reduction, the study found.
The study used official figures -- or estimates when official data was not available -- for deaths from Covid, as well as total excess deaths from each country.
Excess mortality is the difference between the total number of people who died from all causes and the number of deaths expected based on past data.
These analyses were compared with a hypothetical alternative scenario in which no vaccine was administered.
The model accounted for variation in vaccination rates across countries, as well as differences in vaccine effectiveness based on the types of vaccines known to have been primarily used in each country.
China was not included in the study because of its large population and strict containment measures, which would have skewed the results, it said.
The study found that high- and middle-income countries accounted for the largest number of deaths averted, 12.2 million out of 19.8 million, reflecting inequalities in access to vaccines worldwide.
Nearly 600,000 additional deaths could have been prevented if the World Health Organization's (WHO) goal of vaccinating 40 percent of each country's population by the end of 2021 had been met, it concluded.
"Millions of lives have probably been saved by making vaccines available to people around the world," said lead study author Oliver Watson of Imperial College London.
"We could have done more," he said.
Covid has officially killed more than 6.3 million people globally, according to the WHO.
But the organisation said last month the real number could be as high as 15 million, when all direct and indirect causes are accounted for.
The figures are extremely sensitive due to how they reflect on the handling of the crisis by authorities around the world.
The virus is on the rise again in some places, including in Europe, which is seeing a warm-weather resurgence blamed in part on Omicron subvariants.
EU watchdog approves Valneva anti-Covid jab
The EU's medicines watchdog on Thursday approved Valneva's anti-Covid jab, Europe's sixth green-lit coronavirus vaccine, which uses more traditional technology than others on the market which the firm hopes will reduce jab hesitancy.
The French-Austrian drug maker's shot is the sixth vaccine to be approved within the 27-nation bloc and contains inactivated particles of the original coronavirus strain that cannot cause an infection.
"EMA has recommended granting a marketing authorisation for Covid-19 vaccine Valneva for use in the primary vaccination of people from 18 to 50 years of age," the Amsterdam-based agency said.
"After a thorough evaluation, EMA's human medicines committee (CHMP) concluded by consensus that the data on the vaccine were robust and met the EU criteria for efficacy, safety and quality," it added.
The EMA said a study with the vaccine which involved nearly 3,000 people older than 30 showed it triggered the production of higher levels of antibodies against the original SARS-CoV-2 strain than British-Swedish pharma company AstraZeneca's Vaxzevria jab.
But the "proportion of people who produced a high level of antibodies was similar for both vaccines," the EMA said.
The vaccine can be stored at fridge temperature, meaning it can be used in countries where storage at very low temperatures is not available.
The EMA added however that there was limited data on the strength of an immune response against variants of concern, including Omicron subvariants which are currently the dominant strains in many EU countries.
Side effects "were usually mild and cleared within a couple of days after vaccination," the EMA added.
"The most common ones were tenderness or pain at the injection site, tiredness, headache, muscle pain and nausea (feeling sick) or vomiting," it said.
"Based on the available evidence... the benefits of Valneva outweigh its risks," the agency said.
Last month the European Commission said it intended to axe an agreement to buy Valneva's vaccine as it had not yet received marketing authorisation.
The EU's executive arm had signed a deal to buy up to 60 million doses of the VLA2001 vaccine in 2022 and 2023.
Britain approved the Valneva vaccine in April after Bahrain received its first approval for emergency authorisation a month before.
Valneva's vaccine now joins jabs by Pfizer and Moderna, which use messenger RNA technology, AstraZeneca and Johnson & Johnson, which use viral vector technology, and Novavax, which is based on a spike protein produced in a laboratory.
Meanwhile a resurgence of Covid-19 cases in Europe, this time driven by new, fast-spreading Omicron subvariants is casting a pall over summer.
Several Western European nations have recently recorded their highest daily case numbers in months, due in part to Omicron sub-variants BA.4 and BA.5.
The increase in cases has spurred calls for increased vigilance across a continent that has relaxed most if not all coronavirus restrictions.
With inputs from AFP






 Maryam Nawaz can wear uniform!
Maryam Nawaz can wear uniform! Here is all about Madiha Rizvi’s second husband
Here is all about Madiha Rizvi’s second husband Madiha Rizvi ties the knot again
Madiha Rizvi ties the knot again Zara Noor Abbas inspired by Rani Mukerji
Zara Noor Abbas inspired by Rani Mukerji Humayun Saeed and Saboor Aly under fire for close interaction in public
Humayun Saeed and Saboor Aly under fire for close interaction in public Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital
Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital





