Alarm bells as coronavirus cases top 200 in Pakistan
NIH data shows infectivity rate also shooting up to 1.53 : Long wait over as US vaccinates youngest against Covid-19
June 22, 2022 11:03 AM

Alarm bells should be sounded in Pakistan as the number of everyday coronavirus infections has soared to over 200 for the first time in many weeks. Although no death was reported during the last 24 hours (Tuesday) the Covid-19 daily cases have now been building up in Pakistan gradually.
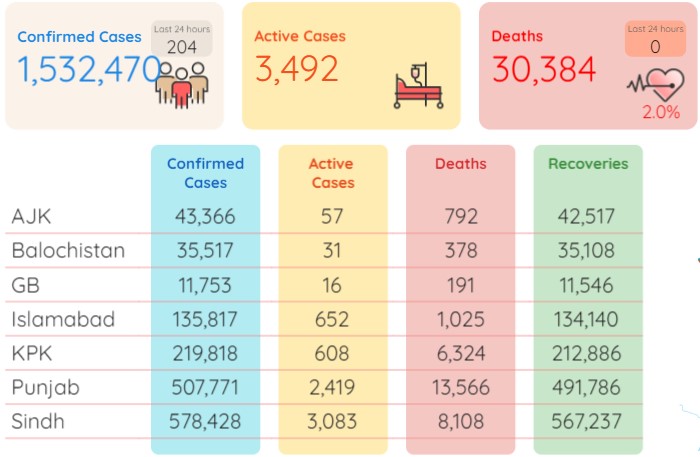
According to the statistics released by the National Institute of Health Pakistan on Wednesday morning, the death toll in Pakistan remained the same at 30,384, whereas the number of total infections now stood at 1,532,470 after adding the fresh 204 cases.
During the last 24 hours (Tuesday), 13,300 tests were conducted throughout Pakistan whereas the positivity ratio also shot up to 1.53 percent. The number of patients in critical care was recorded at 66.
https://twitter.com/NIH_Pakistan/status/1539423844322136064
During the last 24 hours (Tuesday), another 108 people recovered from the Covid-19 in Pakistan and the number of total recoveries now stood at 1,498,594. As of Wednesday, the total count of active cases in the country was recorded at 3,492.
As many as 578,428 coronavirus cases have so far been confirmed in Sindh, 507,771 in Punjab, 219,818 in Khyber Pakhtunkhwa, 135,817 in Islamabad, 35,517 in Balochistan, 43,366 in Azad Kashmir and 11,753 in Gilgit-Baltistan.
As many as 13,566 individuals have lost their lives to the pandemic in Punjab so far, 8,108 in Sindh, 6,324 in KP, 1,025 in Islamabad, 792 in Azad Kashmir, 378 in Balochistan and 191 in Gilgit Baltistan.
US vaccinates youngest against Covid
US hospitals, clinics and pharmacies began vaccinating the nation's youngest children against Covid-19 on Tuesday, a milestone that was welcomed by parents eager to protect kids from the worst impacts of the virus.
Rollout of millions of shots was underway across the country, 18 months after the elderly became the first group eligible for immunization.
Children aged from six months through four years aren't at as great a risk as adults.
But the sheer level of infections has seen more than 45,000 hospitalizations and nearly 500 deaths in the 0-4 group in America since the start of the pandemic -- outcomes that vaccination could have prevented in many cases.
"We're super thrilled," said Amisha Vakil, mother of two three-year-old boys, who wore matching Spiderman tee shirts as they got their Moderna shots at Texas Children's Hospital in Houston.
One of the twins had three open heart surgeries within his first five months.
"He's super high risk so you know, we've been living in a little bubble," said Vakil. "Now he has little armor that helps a lot."
The moment was also hailed by President Joe Biden, whose administration made 10 million shots of the Moderna and Pfizer vaccines available to states after they were authorized last week.
"The United States is now the first country in the world to offer safe and effective Covid-19 Vaccines for children as young as six months old," said Biden, calling it a "monumental step forward."
A handful of other countries and territories including Argentina, Bahrain, Chile, China, Cuba, Hong Kong and Venezuela were previously offering Covid shots for toddlers, but these did not include mRNA vaccines, regarded as the leading technology for the purpose.
The European Medicines Agency is reviewing the Moderna vaccine for use in under-sixes and could follow the US decision.
- Born in pandemic -
Many children being brought in Tuesday were born after the pandemic started and had only known a life of restrictions.
Anna Farrow, who came to the same hospital with her husband Luke, said she saw a new start for their son George, aged three, and Hope, aged 10 months.
"This is sort of the beginning of a regular childhood. And we're very excited about that," she said.
On the other side of the country in Needham, Massachusetts, Ellen Dietrick, an administrator at Temple Beth Shalom was preparing to welcome 300 children on the first day.
Daniel Grieneisen, the father of a three-year-old girl who got the vaccine, said: "It means that we are now just a couple weeks from being able to take her indoors places, and kind of get back to living our lives, it's pretty exciting."
Last week, a panel of experts called by the Food and Drug Administration reviewed data from clinical trials involving thousands of children that were conducted by Pfizer and Moderna, and deemed both of the vaccines safe and effective.
However, a survey carried out by the Kaiser Family Foundation in May found only one-in-five parents of children under five were eager to get them vaccinated right away. A slightly higher proportion, 38 percent, said they would wait and see how well the vaccine worked for others.
New Yorker Rita Saeed, 29, said she was concerned about side effects and planned to wait a couple of years before deciding whether to vaccinate her two-year-old son.
"Each to their own, I think it should be optional, not mandatory," she said, pushing her son in a stroller through Central Park.
Hal Moore, a 32-year-old teacher who lives in New York City, said he was "definitely relieved" that he will be able to vaccinate his 10-month-old daughter Lucy, but "we'll probably wait until her next normal appointment to get it."
In a sign of the ongoing politicization surrounding vaccines in America, Florida governor and possible Republican presidential candidate Ron DeSantis refused to place an order with the federal government for vaccines for the youngest children, leaving private practices and parents to fend for themselves.
"These are the people who have zero risk of getting anything," he said at a press conference last week.
Moderna to build UK centre for mRNA vaccines
Moderna and the UK government on Wednesday announced a deal for the US biotech firm to build a cutting-edge centre to develop and produce mRNA vaccines for respiratory diseases, including Covid.
The company and the health ministry said in a joint statement that work could start on the manufacturing and research and development centre as early as this year.
The first UK-developed mRNA vaccine is scheduled to be produced in 2025.
No financial details or location for the centre were given but ministers said the deal would give patients on the state-run National Health Service access to "next generation" vaccines and treatments.
It would also ensure "consistent home-made supply" and allow increased production in the event of any future health crisis, allowing the hub to be a global base for clinical trials.
"mRNA technology has proven to be one of the fastest routes to develop highly effective vaccines during the pandemic and has been pivotal in protecting people," the health ministry said.
It the technology could lead to breakthrough treatements in a number of disease areas, including cancer, flu, dementia and heart disease.
"Our investment will guarantee jabs in arms against some of the toughest viruses out there, bringing us to the forefront of the fight against future threats," added Prime Minister Boris Johnson.
Britain was one of the worst-hit countries by the Covid outbreak, with nearly 180,000 deaths.
It bought vaccines in bulk even before they were given regulatory approval for use, including Moderna and Pfizer's mRNA jabs.
AstraZeneca working with scientists at the University of Oxford developed their own more conventional vaccine.
With inputs from AFP.






 Maryam Nawaz can wear uniform!
Maryam Nawaz can wear uniform! Here is all about Madiha Rizvi’s second husband
Here is all about Madiha Rizvi’s second husband Madiha Rizvi ties the knot again
Madiha Rizvi ties the knot again Zara Noor Abbas inspired by Rani Mukerji
Zara Noor Abbas inspired by Rani Mukerji Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital
Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital Nazish re-shares fan stand over Babar proposal controversy
Nazish re-shares fan stand over Babar proposal controversy





