Pakistan posts 693 more coronavirus infections, two deaths
NIH data shows almost all indicators moving up slowly: Calls for key Covid drug to be made available outside US: Biden condition has improved since Covid diagnosis
July 23, 2022 10:57 AM

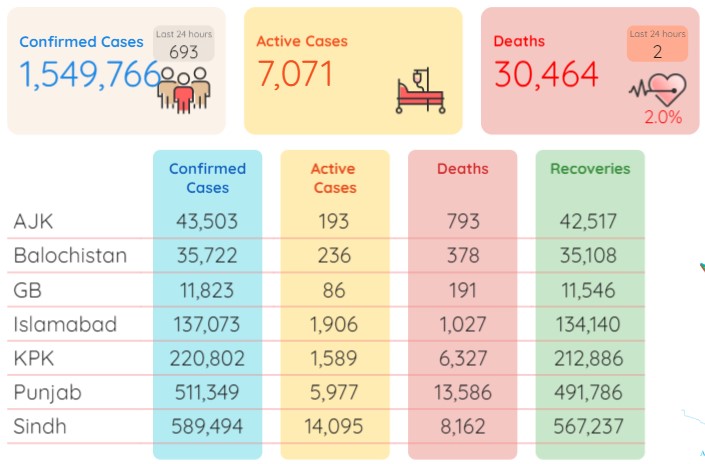
Pakistan has recorded 693 more coronavirus infections and two deaths during the last 24 hours (Friday), showed the statistics released by the National Institute of Health Pakistan on Saturday morning.
As per the NIH data, the death toll in the country now moved up to 30,464 after adding the two new fatalities while the number of total infections now stood at 1,549,766 after adding the fresh 693 cases.
During the last 24 hours (Friday), 23,423 tests were conducted throughout Pakistan whereas the positivity ratio stood at 2.96 percent. The number of patients in critical care was recorded at 180.
The maximum positivity ratio was recorded in Lahore which was at 8.27 percent, followed by Islamabad at 6.50 percent, 5.88 percent in Muzaffarabad, 5.88 percent in Skardu, 4.60 percent in Gilgit, 3.52 percent in Faisalabad, 3.23 percent in Sargodha, 2.92 percent in Peshawar and 2.19 percent in Karachi.
https://twitter.com/NIH_Pakistan/status/1550664045262422016
During the last 24 hours (Friday), another 3,388 patients have recovered from the Covid-19 in Pakistan and the number of total recoveries now stood at 1,512,231. As of Saturday, the total count of active cases in the country was recorded at 7,071.
As many as 589,494 coronavirus cases have so far been confirmed in Sindh, 511,349 in Punjab, 220,802 in Khyber Pakhtunkhwa, 137,073 in Islamabad, 35,722 in Balochistan, 43,503 in Azad Kashmir and 11,823 in Gilgit-Baltistan.
As many as 13,586 individuals have lost their lives to the pandemic in Punjab so far, 8,162 in Sindh, 6,327 in KP, 1,027 in Islamabad, 793 in Azad Kashmir, 378 in Balochistan and 191 in Gilgit Baltistan.
Calls for key Covid drug to be made available outside US
Researchers have called for an experimental Covid treatment, which has shown early indications of being more effective against dominant Omicron subvariants, to be made available outside the United States.
Numerous monoclonal antibody treatments have helped blunt the worst of the disease for patients since the start of the pandemic, but their effectiveness has been weakened as the virus mutates.
However one such anti-viral treatment, bebtelovimab, is "particularly good at targeting the mutated spike protein of the BA.4 and BA.5 Omicron subvariants," Antoine Flahault, director of the Institute of Global Health at the University of Geneva, told AFP.
But as BA.4 and BA.5 run rampant in many countries, the drug made by American pharmaceutical firm Eli Lilly has so far only been made available in US.
Bebtelovimab has not yet been officially approved in the US, but received "emergency use authorisation" earlier this year following positive results from a trial involving patients with mild to moderate Covid symptoms.
The US government ordered 600,000 doses in February, then another 150,000 doses late last month.
But as more evidence emerges of how weak other monoclonal antibodies are against BA.5 and BA.5, some are calling for the rest of the world to get access to the drug.
Five Europe-based medical researchers wrote a letter published in the Lancet Infectious Disease journal this week saying that among monoclonal antibodies, bebtelovimab "stands out" against the dominant Omicron subvariants.
"Bebtelovimab would be an important anti-viral globally," the researchers wrote, adding that it "should be made available outside the USA for patients worldwide".
On Wednesday, France's Covid Scientific Council asked the French government "to do everything possible to ensure that bebtelovimab, the only monoclonal antibody treatment currently active against BA.5, is available as soon as possible in France."
- 'This drug can save lives' -
Eli Lilly told AFP that it "maintains permanent and open communication with health authorities around the world, in order to understand the local need for this antibody, bebtelovimab, which remains experimental for the time being".
The company added that "in Europe, in view of the available data which is considered preliminary," it did not currently plan to make the drug available for trials or apply for authorisation to market it on the continent.
Eli Lilly said bebtelovimab had not been the subject of a European Medicines Agency (EMA) procedure for emergency use, "as was the case for the previous neutralising antibodies against Covid-19."
Given the stakes, some urged quick action.
"This drug can save lives," Flahault said.
"We should consider making it a global public good in order to make it available to very vulnerable patients who could benefit from it around the world."
He said such treatments were particularly important for Covid patients who were at a higher risk of severe symptoms, or whose immune systems had been comprised.
"This is the drug that retains the best efficacy against BA.5," tweeted Renaloo, a French association of kidney disease patients.
"What are our European health authorities waiting for... to guarantee access to immunocompromised patients who need it?"
The calls for wider access come during a pandemic which has consistently seen the wealthy countries where new treatments and vaccines have been developed save the critical first doses for themselves.
The vast gap in access between higher and lower income countries has spurred numerous nations to call for intellectual property rights to be temporarily lifted on Covid vaccines and treatments, allowing them to boost local production.
On Tuesday IFPMA, which represents global pharmaceutical firms, called on wealthy countries to work towards a more equitable distribution for vaccines and treatments in the case of a potential future pandemic.
Biden condition has 'improved' since Covid diagnosis
President Joe Biden's symptoms "have improved" and his key vital signs including blood pressure remain normal, his White House physician said Friday a day after the 79-year-old US leader tested positive for Covid.
Isolating at the White House, Biden has posted on social media several times since his diagnosis that he is feeling "good" and continuing to perform his presidential duties.
"His symptoms have improved," doctor Kevin O'Connor wrote in a memorandum to the White House press secretary.
Although Biden was continuing to experience a runny nose, cough and fatigue, his pulse, blood pressure, respiratory rate and oxygen saturation "remain entirely normal," according to O'Connor.
While his temperature rose slightly Thursday night to 99.4 degrees Fahrenheit (37.4 Celsius), Biden was treated with acetaminophen and his temperature has since remained normal, the physician said.
"The president is tolerating treatment well," including the antiviral pill Paxlovid, used to minimize the severity of Covid-19.
The highly-transmissible Omicron BA.5 subvariant is currently fueling a new Covid wave in the United States -- where hospitalizations have doubled in recent months.
While Biden is reported to be in good general health, as the oldest US president ever elected his age heightens concern over the impact of Covid.
Facing a punishing workload, Biden -- like other presidents before him -- had been showing the strain in recent months: his walk is stiffer, his hair thinner, and his speech less fluent than when he took office.
And politically Biden is in a tough phase of his presidency, facing November midterm elections that are forecast to be painful for his Democratic Party, as well as declining personal approval ratings.
The White House has emphasized since Biden's diagnosis that the president was fully vaccinated and twice boosted -- and O'Connor reiterated that he was expected to "respond favorably" to treatment.
"There has been nothing in the course of his illness thus far which gives me cause to alter that initial expectation," he wrote.
Biden will keep isolating in accordance with guidelines from the US Centers for Disease Control and Prevention, and his medical team will "continue to monitor him closely."
While other members of Biden's family and many close advisors contracted the virus, the president had remained Covid-free until now.
On taking office, he made a point of abiding by strict Covid protocols, holding socially distanced meetings or Zoom gatherings, and wearing a mask to public events -- in sharp contrast to his predecessor Donald Trump.
Then-president Trump tested positive for coronavirus in October 2020 -- in the middle of the bitterly fought election race he eventually lost to Biden.
With inputs from AFP.






 Maryam Nawaz can wear uniform!
Maryam Nawaz can wear uniform! Here is all about Madiha Rizvi’s second husband
Here is all about Madiha Rizvi’s second husband Madiha Rizvi ties the knot again
Madiha Rizvi ties the knot again Zara Noor Abbas inspired by Rani Mukerji
Zara Noor Abbas inspired by Rani Mukerji Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital
Two patients die, injuries of 12 others multiplied after roof collapse at Gujrat hospital Nazish re-shares fan stand over Babar proposal controversy
Nazish re-shares fan stand over Babar proposal controversy





