Coronavirus depleting fast in Pakistan
NCOC data shows only 11 people died, 664 infected during last 24 hours.
October 18, 2021 09:10 AM

Coronavirus pandemic is depleting fast in Pakistan with all the indicators continue to show the downward trend for about two weeks now.
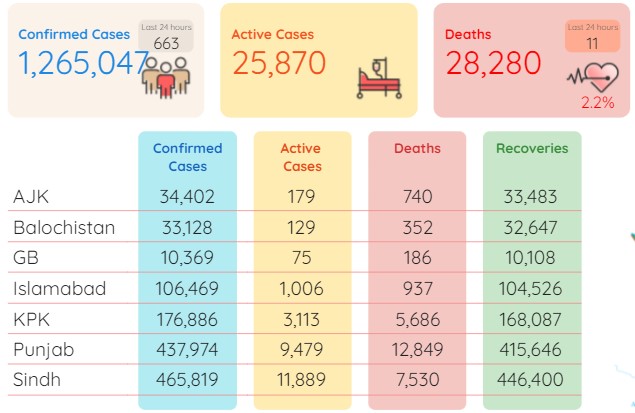
The latest plunge came from the statistics released by the National Command and Operation Centre (NCOC) on Monday morning. During the last 24 hours (Sunday), Pakistan has reported only 11 deaths and 663 infections with the positivity ratio nosediving to just 1.66 percent.
As per the NCOC figures, after the addition of 11 new deaths, the overall toll has now surged to 28,280 whereas the number of total infections now stood at 1,265,047 after adding the fresh 663 cases.
During the last 24 hours (Sunday), a total of 39,902 tests were conducted throughout Pakistan whereas the positivity ratio stood at 1.66 percent. The number of patients in critical care was 1,913.
https://twitter.com/OfficialNcoc/status/1449908773036888066
During the last 24 hours (Sunday), as many as 1,019 patients have recovered from the virus whereas the total recoveries stood at 1,210,897. As of Monday, the total count of active cases in the country was recorded at 25,870.
As many as 465,819 coronavirus cases have so far been confirmed in Sindh, 437,974 in Punjab, 176,886 in Khyber Pakhtunkhwa, 106,469 in Islamabad, 33,128 in Balochistan, 34,402 in Azad Kashmir and 10,369 in Gilgit-Baltistan.
Moreover, 12,849 individuals have lost their lives to the pandemic in Punjab so far, 7,530 in Sindh, 5,686 in KP, 937 in Islamabad, 740 in Azad Kashmir, 352 in Balochistan and 186 in Gilgit Baltistan.
Russia reports record daily virus cases again
Russia on Sunday reported a new record for coronavirus infections for a fourth day in a row, a day after the worst-affected country in Europe set a new death toll high.
There were 34,303 new infections and 997 deaths registered in the country over 24 hours, according to the official toll.
On Saturday, Russia recorded 1,002 deaths -- the first time daily deaths hit the one thousand mark -- and 33,208 new infections.
The climbing Covid-19 statistics come as the nation's vaccination drive is at a standstill and with few restrictions in place.
The Kremlin, which says authorities have to ensure "the economy continues working", has avoided re-introducing major measures despite calling the vaccination rate unacceptably low.
It also insists Russia's medical system is not overwhelmed and could take in the rising number of patients.
Authorities have blamed Russians for the growing outbreak.
Health Minister Mikhail Murashko this week pointed to their "behaviour", while Kremlin spokesman Dmitry Peskov said everything had been done to give the public the chance to "save their lives by getting vaccinated".
While several Russian-developed jabs have been available for months, many people remain vaccine-sceptic.
Independent polls show more than half of Russians do not plan to get a shot.
According to official figures, Russia's fatalities from Covid now stand at 223,312 -- the highest toll in Europe.
Under a broader definition of deaths linked to the virus, the Rosstat statistics agency has said that by the end of August more than 400,000 people in Russia had died with the coronavirus.
US panel votes to authorize J&J booster
A high-level panel of US medical experts voted unanimously Friday to recommend authorizing a second shot of the Johnson & Johnson Covid vaccine.
The decision will help end a sense of limbo for 15 million Americans who received the first dose but were worried whether it was enough to protect them, particularly after the rise of the Delta variant.
The committee members, convened by the Food and Drug Administration (FDA), agreed with the pharmaceutical company that safety and efficacy evidence favored a second dose.
The recommendation may be tweaked further by the FDA and by the Centers for Disease Control and Prevention (CDC), and an emergency use authorization should follow within days to weeks.
Several of the independent experts suggested that though J&J had applied to authorize a booster, a second J&J vaccine should in fact be regarded as an "additional" dose needed to achieve full vaccination.
"I think this frankly was always a two-dose vaccine. It's hard to recommend this as a one-dose vaccine," said Paul Offit, a vaccine expert from Children's Hospital of Philadelphia.
The panel was also asked to discuss, but not to vote on, the question of so-called "mix and match" boosting, in which a person receives a different brand or type of vaccine as a booster than that used for the initial dose.
But FDA scientist Peter Marks said "it's possible" that question will be voted on in future.
A preliminary study this week showed that people who have received the J&J vaccine may benefit from a booster dose of a different, messenger-RNA vaccine such as Pfizer or Moderna.
The single shot J&J vaccine was never as effective as its double-shot mRNA counterparts, but some evidence suggests it is less susceptible to waning than Pfizer and Moderna, because of the viral vector technology behind it.
Nevertheless, the company presented data showing efficacy against symptomatic Covid rose from 70 to 94 percent in a US trial after a second dose given two months after the first, similar to figures associated with mRNA vaccines.
Friday's meeting came after the same panel voted Thursday to authorize a booster dose of Moderna's vaccine for at-risk groups.
In that meeting, the panel decided to authorize a booster dose of Moderna for the over-65s, people aged between 18 and 64 who are at a higher risk of developing a severe version of the coronavirus, and those whose work may involve frequent exposure to the virus.
A similar decision was already reached for the Pfizer vaccine several weeks earlier, with experts concerned a booster might not be suitable for everyone, especially younger males at risk of developing myocarditis (heart inflammation) as a side effect.
But the J&J decision applies more broadly, to everyone over 18 who received the first shot of the vaccine at least two months earlier.
Serious but extremely rare safety concerns include blood clotting with low platelets, particularly in younger women, and Guillain-Barre Syndrome, a rare immune disorder that causes muscle weakness and sometimes temporary paralysis.




 Yet another burden on people: Govt hikes oil prices
Yet another burden on people: Govt hikes oil prices Public reacts to Bohemia's music video ‘Salsa’ starring Sistrology
Public reacts to Bohemia's music video ‘Salsa’ starring Sistrology Saudis offered PIA, airports, joint ventures to build five-star hotels
Saudis offered PIA, airports, joint ventures to build five-star hotels Finance minister rules out further rupee devaluation
Finance minister rules out further rupee devaluation Post-mortem report says woman’s death caused by her falling off train
Post-mortem report says woman’s death caused by her falling off train Hamari Mulaqat: Bushra Ansari opens up about divorce and 2nd marriage to Iqbal Hussain
Hamari Mulaqat: Bushra Ansari opens up about divorce and 2nd marriage to Iqbal Hussain





